Testing of our SERION ELISA agile SARS-CoV-2 IgG
Outstanding study results
In a comparative study from Strömer et al. 2020 the performance of eight different Anti-SARS-CoV-2 IgG immunoassays was analyzed using 26 convalescent sera from PCR-confirmed cases, 27 sera containing neutralizing antibodies against SARS-CoV-2 and 100 pre-pandemic sera from 2018/2019.
Results:
- “Both tests [Virion and Abbott] turned out to be the most sensitive and specific assays in the validation part of the study.”
- “While the N-based IgG assay [Abbott] revealed the existence of two groups of individuals, and one of them showed a rapid decrease of IgG indices within a few months, the IgG reactivity in the S protein-based assay [Virion] was found to be more stable.”
- “This is in-line with a previous preprint (Ripperger et al.) and could suggest that the latter test [Virion] is better suited for epidemiological studies, especially when it comes to questions of prevalence.”
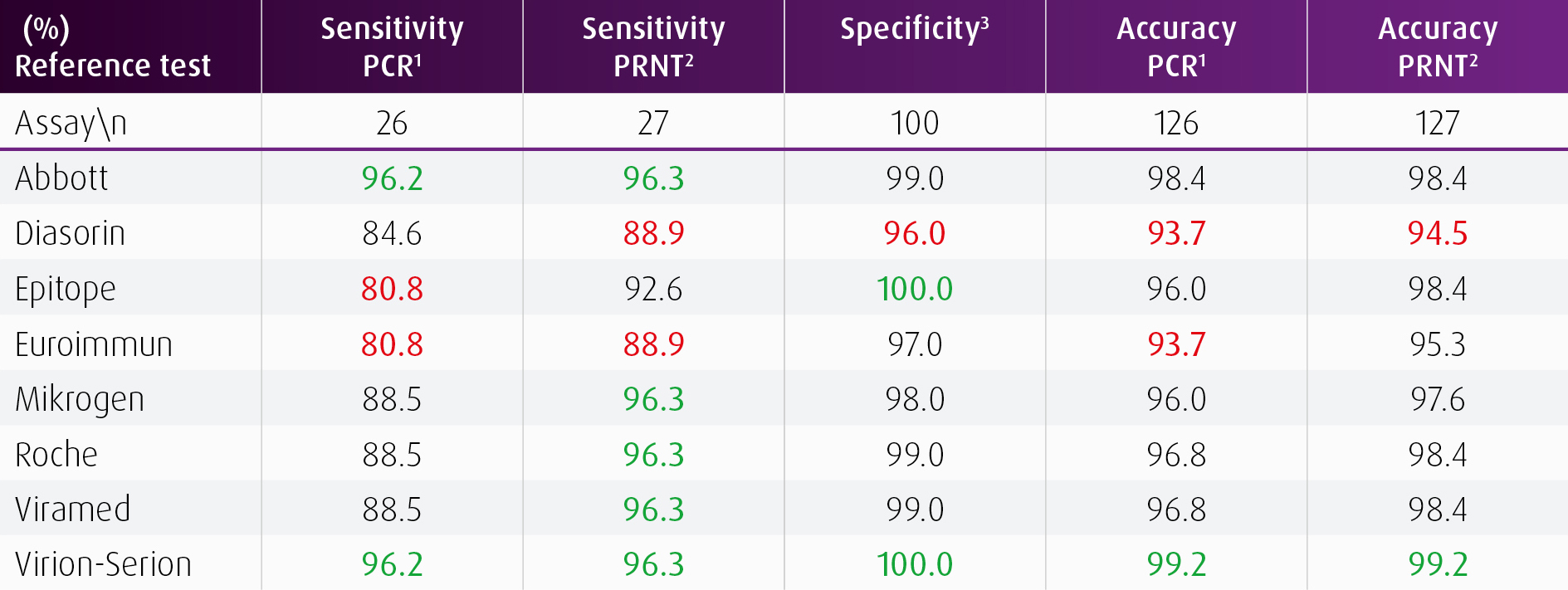
[1] SARS-CoV-2 RNA was previously detected in the PCR test. [2] SARS-CoV-2-neutralizing antibodies with a titer >1:10 were demonstrated in the plaque reduction neutralization test (PRNT). [3] 100 stored sera from 2018/2019
Note: The highest values are shown in green, the lowest values are marked in red.
Click here to go to the study!
Detection of neutralization titers: Highest correlation with SERION ELISA agile SARS-CoV-2 IgG
The publication by Werner et al., 2021 is a comprehensive study evaluating the performance of 16 ELISA/ECLIA-based and 16 LFA-based tests on a defined serum panel. The study also included the analysis of n=22 sera with previously determined SARS-CoV-2 virus neutralization titers. With an R2 of 0.87 the SERION ELISA agile SARS-CoV-2 IgG revealed the best correlation within this evaluation. In general, it has been pointed out that Spike protein based assays showed better correlation with neutralizing antibodies.
Click here to go to the study!
Detection of neutralization antibodies: Highest performance of SERION ELISA agile SARS-CoV-2 IgG
Within the publication of Krone et al., 2021, a comparison of several commercial assays with a neutralization assay was described. A panel of 63 sera has been analyzed with a pseudovirus neutralization assay and in 57 cases neutralizing SARS-CoV-2 antibodies could be detected. These 57 samples positive in the neutralization assay were used to determine sensitivity. Further, specificity was evaluated based on 50 negative sera. SERION ELISA agile SARS-CoV-2 IgG showed the best performance in this comparison, with a sensitivity of 98.3% and a specificity of 100% (Krone et al., 2021).
Click here to go to the study!
Another very recent publication from Perkmann et al., 2021 also included the SERION ELISA agile SARS-CoV-2 IgG assay. The study investigated the immune response from sera of n=69 SARS-CoV-2 naïve individuals after vaccination with BNT162b2 (Pfizer/BioNTech) with five quantitative spike protein based assays.
To analyze their capability to detect neutralizing antibodies, the antibody binding assays were compared to a surrogate virus neutralization test (sVNT) as reference. sVNT assays detect anti-SARS-CoV-2 antibodies, which block the interaction between the receptor binding domain (RBD) of the viral spike glycoprotein with the ACE2 cell surface receptor and are designed to mimic the virus–host interaction in an ELISA format. Since approximately 90% of the neutralizing antibodies that are generated after SARS-CoV-2 infection prevent this interaction, the sVNT is considered a substitute (surrogate) for a conventional neutralization assay.
Overall, a good correlation between SERION ELISA agile SARS-CoV-2 IgG and the sVNT was demonstrated. Especially remarkable is the good correlation with sVNT negative sera. Only in the SERION ELISA agile SARS-CoV-2 IgG the median of antibody activity of sVNT negative samples was borderline. In four other assays a positive median was determined.
Click here to go to the study!
If you have any questions regarding our SARS-CoV-2 Immunoassays or antigens, please feel free to contact our product management:
pm@virion-serion.de
