Antibody tests
SERION ELISA agile
SARS-CoV-2
To be successful in the fight against SARS-CoV-2, it is essential to closely monitor the spread of the virus to enable
- separation of acutely infected from healthy non-infected persons: slow down the pandemic
- pinpointing seroconverted persons: plasma donation for antibody therapy of COVID-19 patients, observation of pandemic development, collection of epidemiological data
To support these goals the SERION ELISA agile SARS-CoV-2 product line offers a unique formulation of antigens that address the different questions.
Antibody tests
SERION ELISA agile
SARS-CoV-2
To be successful in the fight against SARS-CoV-2, it is essential to closely monitor the spread of the virus to enable
- separation of acutely infected from healthy non-infected persons: slow down the pandemic
- pinpointing seroconverted persons: plasma donation for antibody therapy of COVID-19 patients, observation of pandemic development, collection of epidemiological data
To support these goals the SERION ELISA agile SARS-CoV-2 product line offers a unique formulation of antigens that address the different questions.

SERION ELISA agile SARS-COV-2 IgA & IgM
To support the diagnosis of COVID-19 and to supplement direct pathogen detection by qRT-PCR, high sensitivity on the serological test system is required. On the one hand, an early and accurate diagnosis can help to define therapeutic measures correctly and, on the other hand, infected persons can be quarantined in time, which helps to slow down the pandemic. Therefore, SERION ELISA agile SARS-CoV-2 IgA and IgM are based on a mixture of recombinant highly purified nucleocapsid protein and whole spike protein (S1/S2 ectodomain) of SARS-CoV-2, thus mapping the most important immunogenic proteins of SARS-CoV-2.
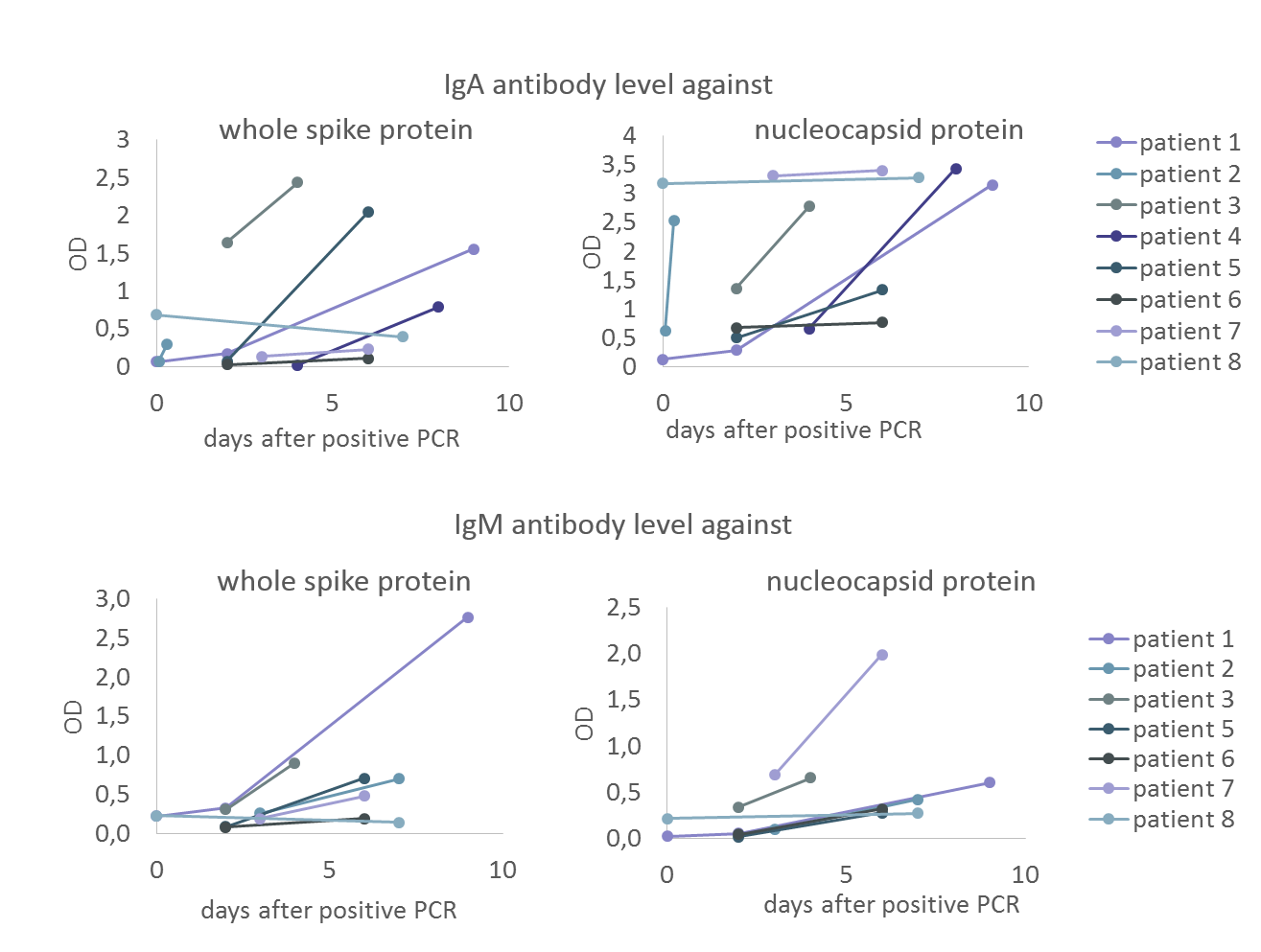
Figure 1: Preliminary data on IgA and IgM antibody levels against SARS-CoV-2 nucleocapsid and whole spike protein over time in acute cases (follow-up sera of patients with positive PCR result). Both anti-nucleocapsid protein and whole spike protein IgA/IgM antibody levels rise in trend.
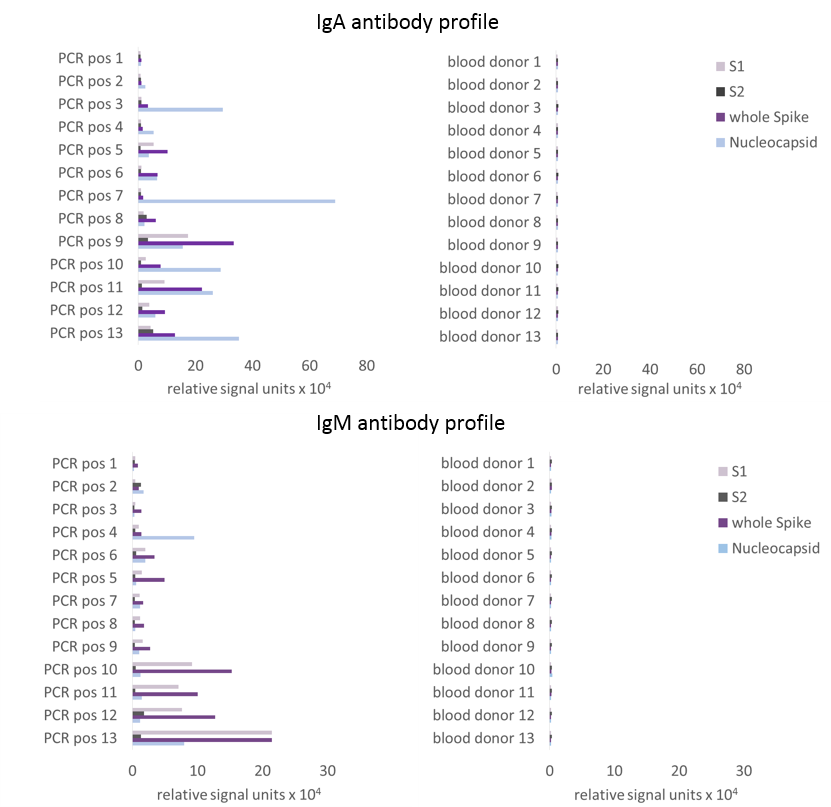
The combined use of the nucleocapsid protein and the whole spike protein ensures high sensitivity (Figure 2)
Figure 2: Preliminary data on the anti-SARS-CoV-2 IgA and IgM antibody profile obtained with a microparticle-based test system (beads were coated with either S1, S2, whole Spike or Nucleocapsid of SARS-CoV-2). The data shown are a representative selection from a total of 337 sera tested (including PCR positive patient sera, sera with suspected infection, blood donor sera, potentially cross-reactive sera) and clearly show that the use of the whole spike protein and the nucleocapsid protein achieves high sensitivities while maintaining high specificities.
SERION ELISA agile SARS-COV-2 IgA & IgM
To support the diagnosis of COVID-19 and to supplement direct pathogen detection by qRT-PCR, high sensitivity on the serological test system is required. On the one hand, an early and accurate diagnosis can help to define therapeutic measures correctly and, on the other hand, infected persons can be quarantined in time, which helps to slow down the pandemic. Therefore, SERION ELISA agile SARS-CoV-2 IgA and IgM are based on a mixture of recombinant highly purified nucleocapsid protein and whole spike protein (S1/S2 ectodomain) of SARS-CoV-2, thus mapping the most important immunogenic proteins of SARS-CoV-2.

Figure 1: Preliminary data on IgA and IgM antibody levels against SARS-CoV-2 nucleocapsid and whole spike protein over time in acute cases (follow-up sera of patients with positive PCR result). Both anti-nucleocapsid protein and whole spike protein IgA/IgM antibody levels rise in trend.
The combined use of the nucleocapsid protein and the whole spike protein ensures high sensitivity (Figure 2)
Figure 2: Preliminary data on the anti-SARS-CoV-2 IgA and IgM antibody profile obtained with a microparticle-based test system (beads were coated with either S1, S2, whole Spike or Nucleocapsid of SARS-CoV-2). The data shown are a representative selection from a total of 337 sera tested (including PCR positive patient sera, sera with suspected infection, blood donor sera, potentially cross-reactive sera) and clearly show that the use of the whole spike protein and the nucleocapsid protein achieves high sensitivities while maintaining high specificities.
SERION ELISA agile SARS-COV-2 IgG
In contrast to acute diagnostics, the detection of anti-SARS-CoV-2 IgG antibodies for pathogen contact confirmation, requires a high specificity of the serological test system. False-positive results would suggest a possible immunity, which not only has a negative effect on the individual but also on the social level (spread of the pandemic). Since immunity is mediated by protective antibodies, which in case of SARS-CoV-2 IgG are mainly formed against the spike protein, the SERION ELISA agile SARS-CoV-2 IgG is based exclusively on the whole spike protein (S1/S2 ectodomain). The use of the whole spike protein compared to a single protein domain (S1, S2 or RBD) increases the sensitivity of the test while maintaining the same specificity (Figure 3).
This specific test setup also offers the possibility of screening for potential donors with protective antibodies whose plasma can be used for therapeutic purposes.
Of course, antigens are used that are produced in house under controlled conditions. Recombinant expression in eukaryotic cells comes closet to natural conditions and high batch comparability and constant availability is guaranteed by expression in bioreactor systems.
Figure 3: Preliminary data on the anti-SARS-CoV-2 IgG antibody profile obtained with a microparticle-based test system (beads were coated with either S1, S2 or whole Spike). The data shown are a representative selection from a total of 337 sera tested (including PCR positive patient sera, sera with suspected infection, blood donor sera, potentially cross-reactive sera) and clearly show that the whole spike protein leads to increased sensitivity while maintaining the same specificity.
Highlights IgA, IgM & IgG:
- Sensitive IgM and IgA Assay as an ideal supplement to direct pathogen detection by combined use of nucleocapsid and whole spike protein
- Speficic IgG assay by exclusive use of the whole spike protein for various applications: determination of immune status, collection of epidemiological data or data on pandemic development, screening of plasma donors for therapeutic purposes
- In house produced antigens guaranteed reliable and reproducible results and sufficient production quantities
- CE-marked assays to support in the diagnosis of COVID-19
For questions regarding our antibody tests please contact our product management: pm@virion-serion.de